
When <<Km, almost all enzyme will be present as. Most in vivo enzymes are exposed to that are less than or equal to Km. Adding more SS cannot increase the rate because there are no unoccupied active sites, in other words, ≈0. At Vmax, the reaction is independent of (zero order). Therefore, /≈1 and V0/Vmax is also ≈1, and a maximum rate will occur. When >Km, almost all enzyme will be present as ES. Since half of Etotal is in the ES form, the other half must be present as Efree, therefore =. 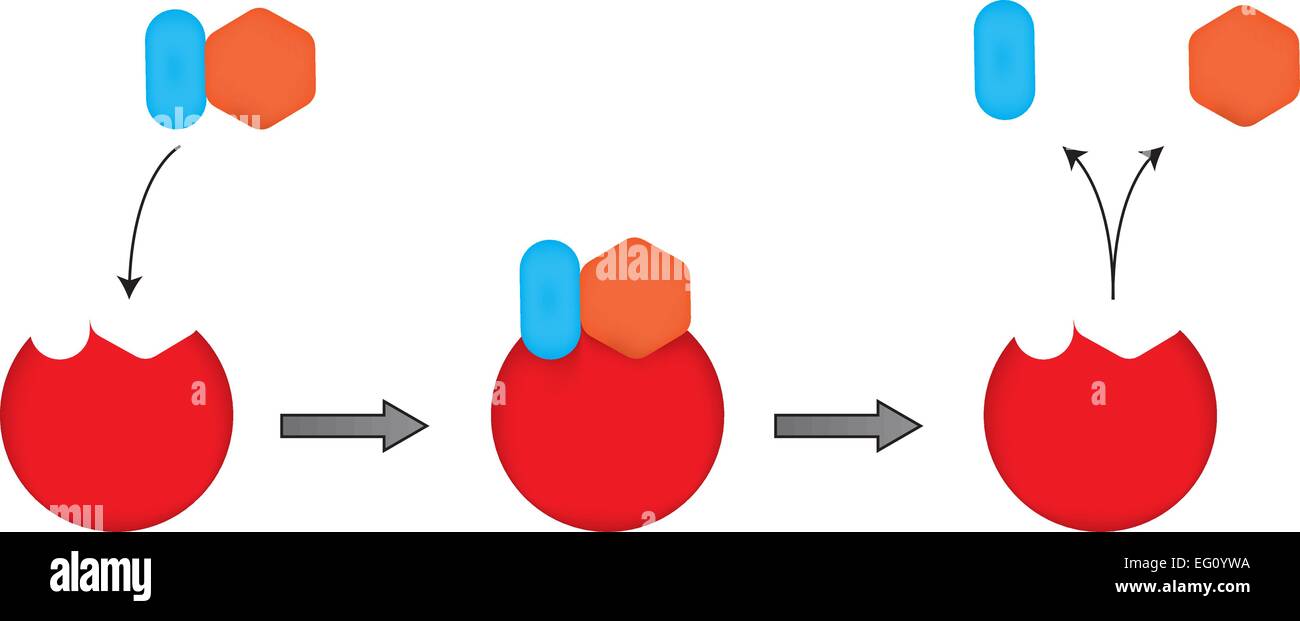
Since V0 is determined by the amount of ES complex formed, a velocity of Vmax/2 will occur when half of the active sites are filled, or, =/2. Similarly, inhibitor E decreases the Vmax of carboxypeptidase A.įor the condition =Km, Km is defined as the that results in half of the maximum velocity (Vmax), i.e., V0=Vmax/2. The data show that addition of inhibitor B results in a decrease in the Vmax of chymotrypsin. Inhibitor B and inhibitor E are noncompetitive inhibitors. In either case, the result is a decrease in the enzyme's Vmax and no change to its KM. Noncompetitive inhibitors, on the other hand, bind to either an allosteric site on the enzyme or to the enzyme-substrate complex. The data show that inhibitor C decreases both the KM and Vmax of penicillinase, making it an uncompetitive inhibitor. Thus, uncompetitive inhibitors both decrease the Vmax of an enzyme and decrease its KM. Conversely, uncompetitive inhibitors bind to the enzyme-substrate complex, thereby decreasing an enzyme's maximal rate of catalysis and increasing its affinity for its substrate. Likewise, inhibitor D increases the KM of lysozyme. The data show that addition of inhibitor A results in an increase in the KM of carbonic anhydrase. Inhibitor A and inhibitor D are competitive inhibitors. Competitive inhibitors do not change an enzyme's Vmax. In other words, competitive inhibitors result in an increase in the KM value of an enzyme for its substrate. Competitive inhibitors bind directly to an enzyme's active site and, in doing so, decrease the affinity of an enzyme for its substrate. The KM of an enzyme is the concentration of substrate required for an enzyme to reach half of its Vmax. 
The Vmax of an enzyme is the rate at which an enzymatic reaction proceeds when the enzyme is saturated with substrate. Two of the primary kinetic parameters in enzymatics are Vmax and KM. Since this value depends upon the reactants and products and not the transition state, enzymes do not affect it.Įnzyme inhibitors affect one or more kinetic parameters associated with catalysis. The free energy of a reaction determines the spontaneity of a reaction. Enzymes also cannot change the value of ΔG for a reaction. Equilibrium depends on the free‑energy difference between the reactants and products, or ΔG, for the reaction. Enzymes cannot change the position of equilibrium rather, they assist in reaching equilibrium. A lower activation energy will increase the magnitude of the rate constant, which in turn means a faster reaction. Activation energy and rate constant are inversely and exponentially related. Enzymes lower the activation energy, thereby promoting the formation of the high‑energy transition state. The activation energy is the energy difference between the transition state and the reactants. Enzymes also lower the activation energy for a reaction. Transition states are in‑between molecules that are not quite the reactants, but also not yet the final product or products. These conformations are called transition states. As a reaction proceeds, the reactants often enter into strained conformations that have much higher free energy values than the reactants and products. Enzymes promote the formation of the transition state. Enzymes promote the formation of a transition state, lower the activation energy, and increase the magnitude of the rate constant for both the forward and reverse reactions, as the enzyme catalyzes both reactions. Enzymes are biological catalysts that increase the rates of chemical reactions without being transformed in the process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
